Introduction

Fipronil is a key active ingredient in many veterinary spot-on treatments, but it is highly sensitive to conditions during production. Exposure to oxygen, heat, and prolonged time in the filling system can cause degradation, reducing potency, shortening shelf life, and creating compliance issues before the product reaches veterinarians or pet owners.
A reliable filling machine manufacturer understands these challenges. Nitrogen purging on a fipronil filling line is not optional: it is essential to protect the formulation. This article explains the chemistry behind active ingredient degradation, how typical filling processes can trigger it, and the critical design features a nitrogen-protected filling system must include to maintain stability from tank to sealed pipette.
Why Active Ingredient Stability Matters in Veterinary Spot-On Products
The Sensitivity of Fipronil and Similar Actives
According to PubMed’s review of fipronil toxicology and metabolism, fipronil is widely used as a broad-spectrum phenylpyrazole insecticide and veterinary drug. Research confirms that oxidative stress is a primary mechanism affecting its stability, with various CYP450 enzymes involved in its metabolic transformation and oxidative degradation pathways.

In simpler production terms, this means that when fipronil is exposed to oxygen during the filling process, the same oxidative reactions that degrade the molecule in biological systems begin to occur in the formulation itself. The active ingredient converts to its degradation products, including fipronil-sulfone and fipronil-desulfinyl, which carry different toxicological and efficacy profiles than the parent compound.
For a manufacturer, this means that a batch filled without inert gas protection may contain less active fipronil than the label claims, even before the product leaves the production facility.
Risks of Degradation During Filling
According to Katyayani Organics, under natural or biotic processes, fipronil is mainly dissipated by photolysis, hydrolysis, reduction and oxidation into four degradation compounds: fipronil-desulfinyl, fipronil-amide, fipronil-sulfide, and fipronil-sulfone.
Of these pathways, oxidation is the one most directly controllable during the filling process. Photolysis can be managed through amber pipette materials and light-controlled production areas. Hydrolysis is less relevant in oil-based spot-on formulations. Oxidation, however, occurs continuously whenever the formulation is in contact with oxygen-containing air, and the filling process creates multiple points of oxygen exposure.
These points include the headspace above the formulation in the supply tank, the air displaced by the liquid as it moves through the pipeline, the moment of filling when the open pipette is in contact with the fill environment, and the residual air in the container headspace before sealing.
The Need for Controlled Filling Environments
A nitrogen purging filling line addresses all of these exposure points by replacing oxygen with inert nitrogen gas at every stage where the formulation contacts the fill environment. The result is a production process where the formulation is never in contact with oxygen from tank to sealed container.
According to MVS Engineering’s pharmaceutical nitrogen application review, for drug products prone to oxidation, nitrogen is used to replace the air in the void space before sealing, reducing the effect of oxygen or moisture and increasing the stability and shelf life of the drug product.
Recommended Reading: Handling Viscosity: Optimizing the Filling of Oil-Based Anti-Parasitics for Pets – King Pack – King Pack Machinery
How Liquid Filling and Packaging Equipment Affect Fipronil Stability
Oxidation from Air Exposure

Oxygen is present in ambient air at approximately 21%. Every time the surface of an oil-based veterinary formulation contacts air during the filling process, the dissolved oxygen in that air begins to react with the active ingredient and the carrier solvent.
In an oil-based spot-on filling line, these contact events happen at the tank surface, at every pipeline connection point that is not fully sealed, and inside each pipette during the fill stroke.
The rate of oxidative degradation depends on temperature, the specific chemistry of the active ingredient and solvent, and the concentration of dissolved oxygen in the formulation. For fipronil in an oil-based carrier, the combination of ambient temperature production conditions and unprotected air contact creates a meaningful degradation risk over the course of a production run.
Temperature and Process Instability
Temperature affects both the rate of oxidative degradation and the viscosity of the oil-based carrier. Higher temperatures accelerate the chemical reactions that drive fipronil degradation. They also reduce formulation viscosity, which changes the flow behavior at the filling nozzle and can affect fill volume accuracy.
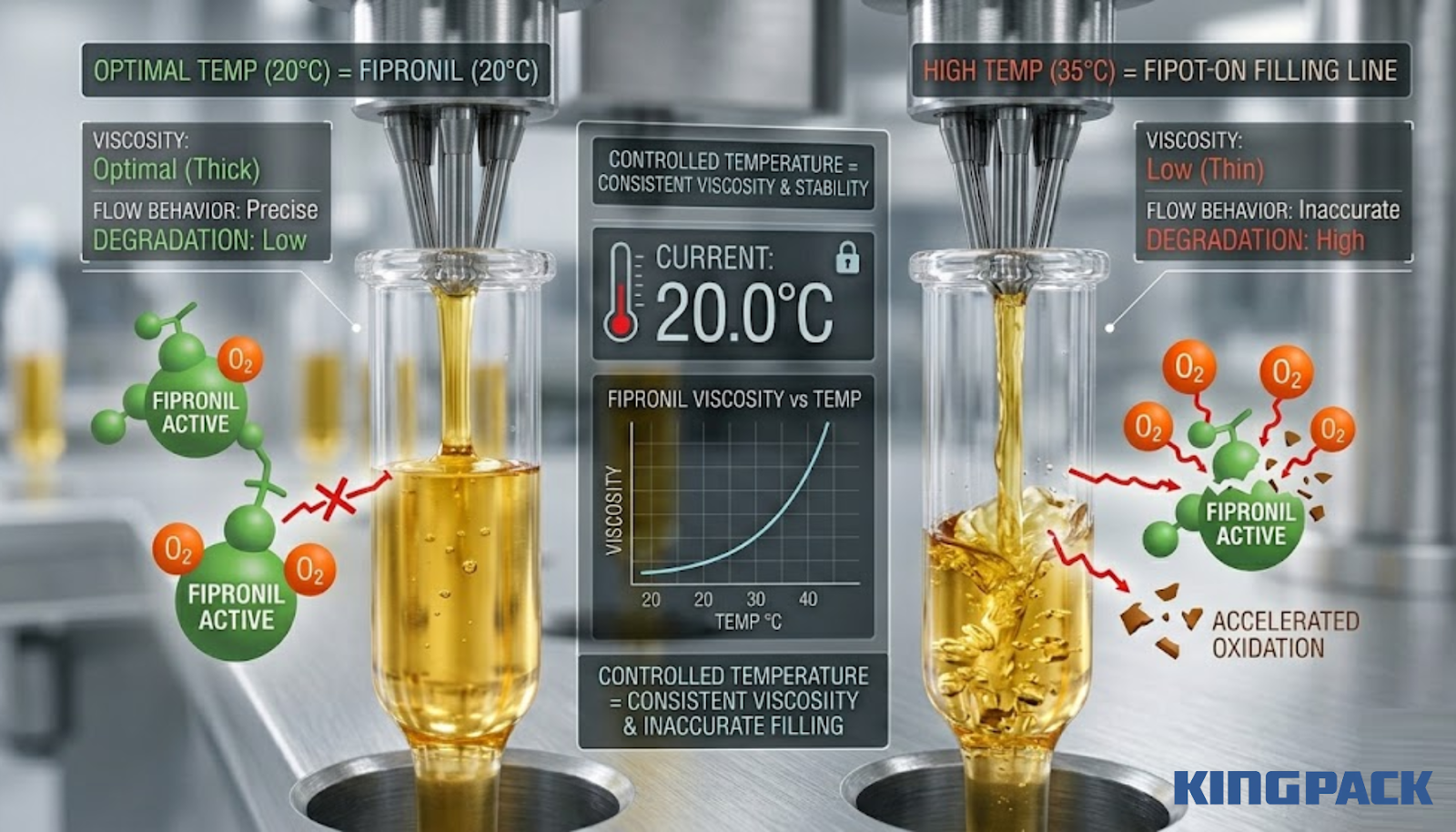
In a production environment where ambient temperature varies between shifts or between seasons, an uncontrolled formulation temperature creates two simultaneous problems: accelerated degradation and inconsistent filling accuracy. Both are managed through the same temperature-control system that maintains constant viscosity for the filling process.
Extended Residence Time in Filling Systems
Every minute the formulation spends in the filling system before it is sealed into a pipette is a minute of potential degradation. Dead volume in the pipeline, long distances between the supply tank and the filling head, and slow production speeds that extend batch duration all increase the total residence time of the formulation in the system.
This is why nitrogen purging on a fipronil filling machine cannot be addressed in isolation from the filling system design. Minimizing dead volume, using short direct pipelines, and maintaining a continuous inert atmosphere throughout the system are all part of the same contamination control strategy.
Recommended Reading: No More Overflow: Bottom-Up Filling Technology for High-Foam – King Pack Machinery
Nitrogen Purging in Automatic Liquid Filling Machines: What is it?
Basic Principle of Nitrogen Protection
Nitrogen is chemically inert under normal production conditions. It does not react with fipronil, oil-based carriers, or any of the active ingredients commonly used in veterinary antiparasitic formulations. When it replaces oxygen in the space above the formulation and inside the container headspace, the oxidative degradation pathway is removed.

According to ON2Quest’s nitrogen purging and blanketing review, nitrogen blanketing, also known as inerting, maintains an inert gas at the surface layer of a product during manufacturing, in storage tanks, or containers, protecting the product from oxidation, degradation, and microbial growth by preventing the ingress of oxygen and moisture.
In a nitrogen protection filling system for veterinary spot-on production, nitrogen is not just applied at one point. It is maintained continuously across the entire product contact pathway.
Where Nitrogen Is Applied in the Process
A complete nitrogen-protected filling line applies inert gas protection at four distinct points:
- Tank blanketing, where nitrogen is maintained above the formulation surface in the supply tank throughout the production run
- Pipeline inerting, where nitrogen purges the wetted pathway before the formulation is introduced and maintains positive pressure in the headspace of sealed pipeline sections
- Nozzle zone protection, where a local nitrogen curtain around the filling nozzle displaces ambient air from the immediate fill environment during each fill stroke
- Container headspace replacement, where nitrogen is injected into the pipette or ampoule immediately before the cap is applied, replacing the residual air with inert gas before sealing
| Protection Point | Mechanism | Benefit |
| Supply tank | Nitrogen blanket above liquid surface | Prevents surface oxidation throughout the batch |
| Pipeline | Sealed system with nitrogen headspace | Eliminates oxygen ingress at connection points |
| Fill zone | Nitrogen curtain at nozzle | Removes ambient air from open container at fill moment |
| Container headspace | Pre-seal nitrogen injection | Seals inert atmosphere inside each pipette |
Difference Between Standard Filling and Nitrogen-Protected Filling
In a standard spot-on filling machine without inert gas protection, the formulation surface in the supply tank is in direct contact with air. The pipeline has connection points where air can enter. The open pipette contacts the fill room atmosphere during the fill stroke. The headspace above the fill level inside the pipette contains air when the cap is applied.
In a nitrogen-protected filling line, each of these contact events is eliminated. The formulation is sealed from air contact at every stage of the process, from tank to sealed pipette.
How Nitrogen Purging Protects Fipronil Formulations
Reducing Oxygen Exposure
The measurable target for nitrogen purging effectiveness in a fipronil filling line is the residual oxygen concentration inside the sealed container after filling. A standard fill in ambient air seals approximately 21% oxygen in the container headspace.
A nitrogen-protected fill using acasi machinery, rotary fillers, or bottle unscramblers targets residual oxygen below 1%, often below 0.5% with optimized purging parameters.
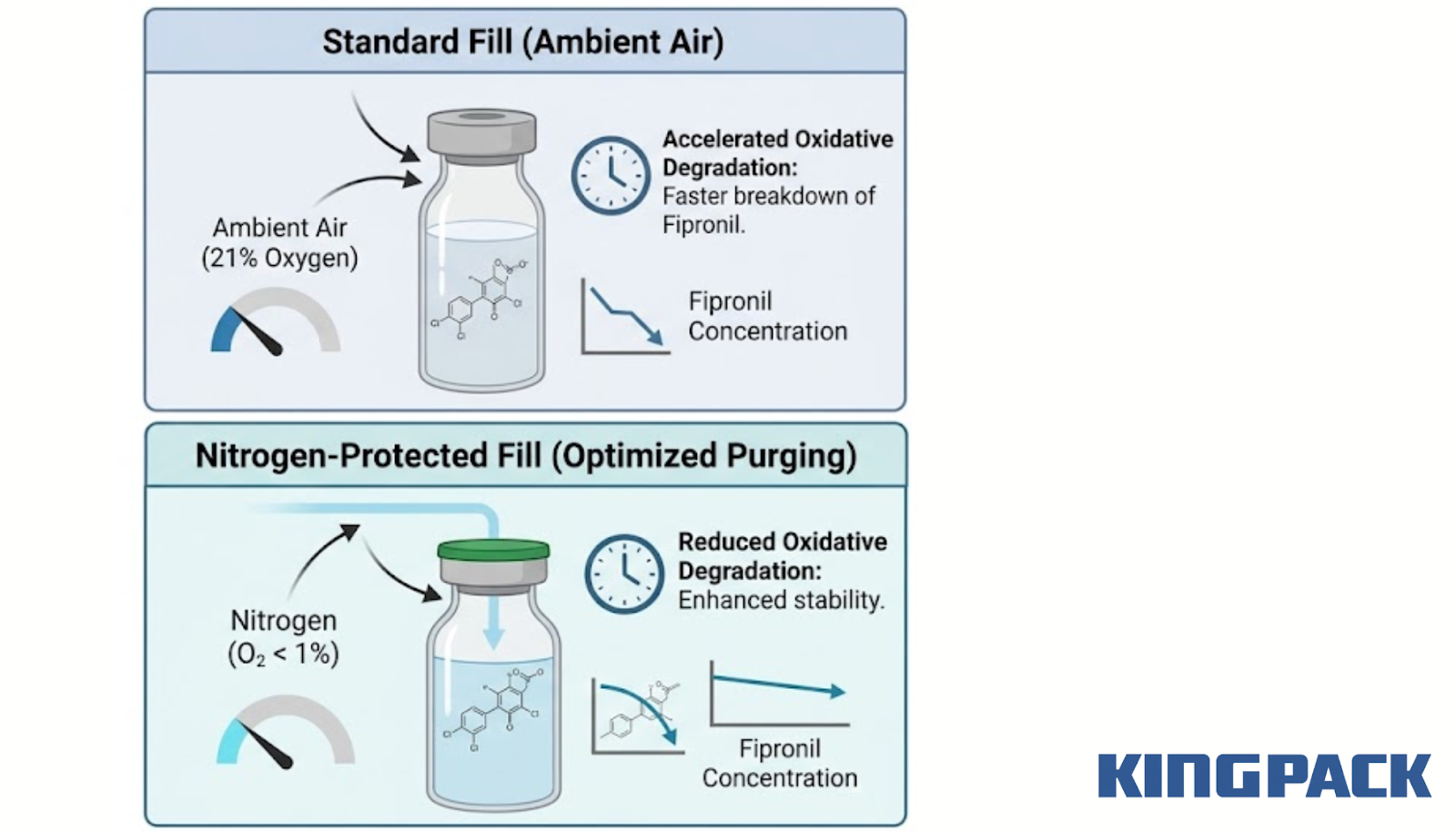
That reduction in headspace oxygen concentration directly reduces the rate of oxidative degradation during storage. Precision control in bottling machinery and packaging solutions further limits human error, ensuring the formulation maintains its active ingredient concentration at or above the label claim throughout the stated shelf life.
Maintaining Chemical Stability
Maintaining active ingredient stability across the full shelf life of a veterinary spot-on product is a regulatory requirement, not just a quality preference. Stability data submitted with the product registration dossier must demonstrate that the active ingredient concentration remains within specification at defined time points under defined storage conditions.
If the filling process introduces oxygen exposure that was not present in the stability study batches, the commercial product may degrade faster than the stability data predicts. Using trusted partner companies and machinery with precision control, technical support, and spare parts minimizes risk of human error. First batches may pass, while later batches without consistent nitrogen protection could fail stability retest.
Extending Shelf Life
According to Springer’s pharmaceutical packaging and stability review, nitrogen in pharmaceutical packaging significantly reduces oxidative degradation. Studies show products packaged under nitrogen maintain active ingredient concentrations above specification for periods two to three times longer than equivalent products packaged in air under the same storage conditions.
For a veterinary spot-on manufacturer, extending shelf life from 18 months to 36 months through nitrogen-protected liquid filling equipment and packaging solutions reduces the risk of product expiry in the distribution channel. Ongoing support, customization options, and innovative solutions across various industries, including beverage and veterinary, help companies maintain reliability and precision while meeting specific needs.
Recommended Reading: Explosion-Proof Requirements for Pet External Sprays: Safe Filling for Alcohol-Based Formulations – King Pack – King Pack Machinery
Key Design Points for Nitrogen-Protected Filling Lines
Sealed Material Tanks with Nitrogen Blanketing
The supply tank is where nitrogen protection begins. The tank must be fully sealed, with a nitrogen inlet valve, a pressure relief vent, and a continuous nitrogen supply maintaining positive pressure above the formulation surface throughout the production run.
The nitrogen pressure in the tank headspace is typically maintained at 0.02 to 0.05 bar above atmospheric pressure. This positive pressure prevents ambient air from entering through any seal or connection point on the tank. A pressure gauge and low-pressure alarm on the nitrogen supply circuit alert the operator if the nitrogen supply falls below the minimum required pressure.
Nitrogen Injection at Filling Nozzles
The filling nozzle zone is the most critical point of oxygen exposure in the entire filling process. At the moment the pipette is open and the nozzle is dispensing the formulation, the container interior is briefly exposed to the fill room atmosphere if no local protection is applied.
A nitrogen curtain at the nozzle creates a localized inert atmosphere around the nozzle tip and the open container during the fill stroke. The nitrogen flow rate is set to displace ambient air from the immediate fill zone without creating turbulence that would disturb the liquid surface inside the pipette.
Our fipronil filling machine configurations at King Pack integrate nitrogen curtain nozzles as a standard design element in our spot-on filling line systems, ensuring that the formulation is protected at the highest-risk point of the process.
Controlled Headspace Nitrogen Replacement
Immediately before the cap is applied to each filled pipette or ampoule, a brief nitrogen injection into the container headspace displaces the residual air above the liquid surface. This step reduces the residual oxygen in the sealed container to the target level, typically below 1%.
The nitrogen injection is timed to the capping cycle and controlled by the line PLC. Injection duration, flow rate, and timing relative to the cap application are all set as parameters in the product recipe and validated as part of the filling line qualification.
Integrated Oxygen Monitoring Systems
Real-time oxygen monitoring in the fill zone and inside sealed containers provides continuous verification that the nitrogen protection system is functioning as designed. Fixed oxygen sensors in the fill zone measure the ambient oxygen concentration around the nozzle during production. Headspace oxygen analyzers on a sampling basis verify the residual oxygen level inside sealed pipettes from each batch.
If the fill zone oxygen concentration rises above the defined alert limit, the monitoring system triggers an alarm and, in an interlock-configured system, stops the filling line until the nitrogen supply is confirmed and the oxygen concentration returns to specification.
Application in Oil-Based Veterinary Anti-Parasitic Products
Characteristics of Oil-Based Spot-On Formulations
Oil-based spot-on formulations for flea and tick prevention present two specific challenges for nitrogen protection filling systems. First, the oil carrier dissolves oxygen more readily than aqueous solutions, meaning that oxygen absorbed into the oil during any unprotected process step is more difficult to remove than surface-level air exposure. Second, the viscosity of oil-based carriers means that the formulation moves slowly through the pipeline, increasing residence time and potential oxidation exposure at each contact point.
Both of these characteristics make nitrogen protection more important for oil-based spot-on products than for aqueous veterinary liquid formulations. The combination of high dissolved oxygen capacity and slow movement through the filling system creates a higher cumulative oxidation risk per batch.
Common Packaging Formats
The two primary packaging formats for fipronil-based veterinary spot-on products, single-dose plastic pipettes and small glass ampoules, have different nitrogen protection requirements at the headspace sealing step.
| Format | Headspace Volume | Nitrogen Injection Timing | Capping Method |
| Plastic pipette (0.5 ml to 5 ml) | Small, 0.1 to 0.5 ml | Pre-cap, brief pulse | Twist-off cap or heat seal |
| Glass ampoule (1 ml to 10 ml) | Moderate, 0.2 to 1 ml | Pre-stopper, continuous flow | Stopper and crimp cap |
| Dropper bottle (5 ml to 30 ml) | Larger, 1 to 5 ml | Pre-dropper insertion | Dropper and screw cap |
For plastic pipettes specifically, the small headspace volume means that a short, precisely timed nitrogen injection is sufficient to achieve the target residual oxygen level. For larger glass formats, a longer continuous nitrogen flow during the filling stroke itself may be needed to adequately displace air from the larger headspace volume before sealing.
Importance of Consistent Dosage and Stability
A fipronil spot-on product that delivers a consistent, stable dose of active ingredient at the point of use is the foundation of the prescribing veterinarian’s confidence in the product. If the filling process allows even partial degradation of the active ingredient between manufacture and use, the clinical performance of the product in the field becomes unpredictable.
Nitrogen purging on the filling line is one of the few process controls that simultaneously protects product efficacy, supports regulatory compliance, and protects the manufacturer’s market reputation. All three outcomes depend on the same engineering decision made at the filling line design stage.
Recommended Reading: Cross-Contamination Prevention: The Gold Standard for Multi-Formula Pet Drop Production – King Pack – King Pack Machinery
Labeling Machines and Packaging Equipment for GMP Compliance
GMP Requirements for Veterinary Pharmaceuticals
GMP guidelines for veterinary pharmaceutical production require manufacturers to demonstrate that their production process does not adversely affect product quality.
For oxygen-sensitive formulations like fipronil-based spot-ons, this means the filling process must be validated to demonstrate that active ingredient degradation during filling is controlled to below a defined limit.
The validation documentation for a nitrogen-protected filling line covers the nitrogen supply system qualification, headspace oxygen measurement validation, and the correlation between fill zone oxygen concentration and active ingredient stability in the sealed container.
Controlled Environment and Clean Production
Nitrogen protection on a spot-on filling line operates within the same GMP production environment requirements that govern all other aspects of veterinary liquid filling. Temperature control, personnel hygiene, equipment cleaning, and batch documentation all continue to apply alongside the inert gas protection system.
The nitrogen supply system itself must be maintained and calibrated as part of the line’s ongoing GMP maintenance program. Nitrogen purity certification, regulator calibration records, and supply pressure logs are all part of the quality documentation package for a nitrogen-protected filling batch.
Documentation and Traceability
Every parameter of the nitrogen protection system operating during a production batch is recorded in the electronic batch record alongside the filling parameters. Nitrogen supply pressure, fill zone oxygen concentration, headspace oxygen results from sampling, and any alarms or deviations are all captured and linked to the batch record. This provides the traceability required for batch release and supports any stability investigation that references the production conditions of a specific batch.
Common Challenges and How to Solve Them
Nitrogen Consumption Optimization
Nitrogen is not free. In a high-speed spot-on production line using automatic machines, liquid fillers, or piston fillers across multiple shifts, nitrogen consumption for tank blanketing, nozzle curtains, and headspace injection can be a significant operating cost.
Maintaining optimal efficiency requires setting and validating the minimum flow rate that achieves the target headspace oxygen concentration. Using excess nitrogen increases cost without improving protection. Optimized nitrogen use helps reduce waste while keeping formulations stable.
System Sealing and Leak Prevention
The effectiveness of nitrogen protection depends on the sealing integrity of packaging machines, capping machines, and other liquid filling equipment. Any leak in tanks, pipelines, or nozzle assemblies allows ambient air to contact the formulation, increasing degradation risk.
Regular leak testing of all nitrogen-protected circuit components is critical. Our designs use fully sealed tri-clamp connections and PTFE gaskets to maintain integrity with oil-based solvents, even during extended production runs.
Compatibility with Existing Production Lines
Manufacturers with existing spot-on filling lines can integrate nitrogen protection without replacing the core filling equipment in most cases. The nitrogen blanketing system on the supply tank, the nozzle curtain, and the headspace injection module can each be retrofitted as independent additions to an existing line, with the control parameters integrated into the existing PLC system.
We provide retrofit nitrogen purging system designs for existing fipronil filling machine configurations as part of our engineering support services at King Pack, allowing manufacturers to upgrade their inert gas protection without a full line replacement.
Recommended Reading: How to Achieve ±1% Filling Accuracy for High-Value Pet Spot-on Treatments – King Pack Machinery
Why Use Global Leader King Pack Nitrogen-Protected Filling Solutions?
At King Pack, our nitrogen purging filling line designs for veterinary spot-on production integrate inert gas protection at every product contact point as a standard engineering feature, not an add-on.
Our pharmaceutical and medical filling lines for fipronil and oil-based anti-parasitic products combine servo-driven piston filling at ±1% accuracy with fully sealed nitrogen-blanketed supply systems, nozzle curtain protection, and headspace replacement before sealing.
The entire system is designed for GMP-compliant production with full electronic batch recording and oxygen monitoring integration.
Contact King Pack to discuss a customized nitrogen-protected filling line solution for your veterinary antiparasitic production requirements.

