Introduction

In pharmaceutical industry, sterile pet pharmaceutical drops sit at the most technically demanding end of veterinary liquid manufacturing. Ophthalmic, otic, and nasal formulations for animals carry the same contamination risk as their human pharmaceutical equivalents, and regulatory bodies treat them accordingly.
In this article, we cover the full architecture of a GMP Filling Equipment Line for sterile pet pharmaceutical drops. We explain the cleanroom grading requirements, the sterile filling process, contamination control strategy, and the precision dosing technology required to meet both veterinary GMP standards and consistent fill accuracy across production.
Why Sterile Standards Are Critical in Veterinary Drop Manufacturing Filling System
Different Types of Sterile Pet Drops
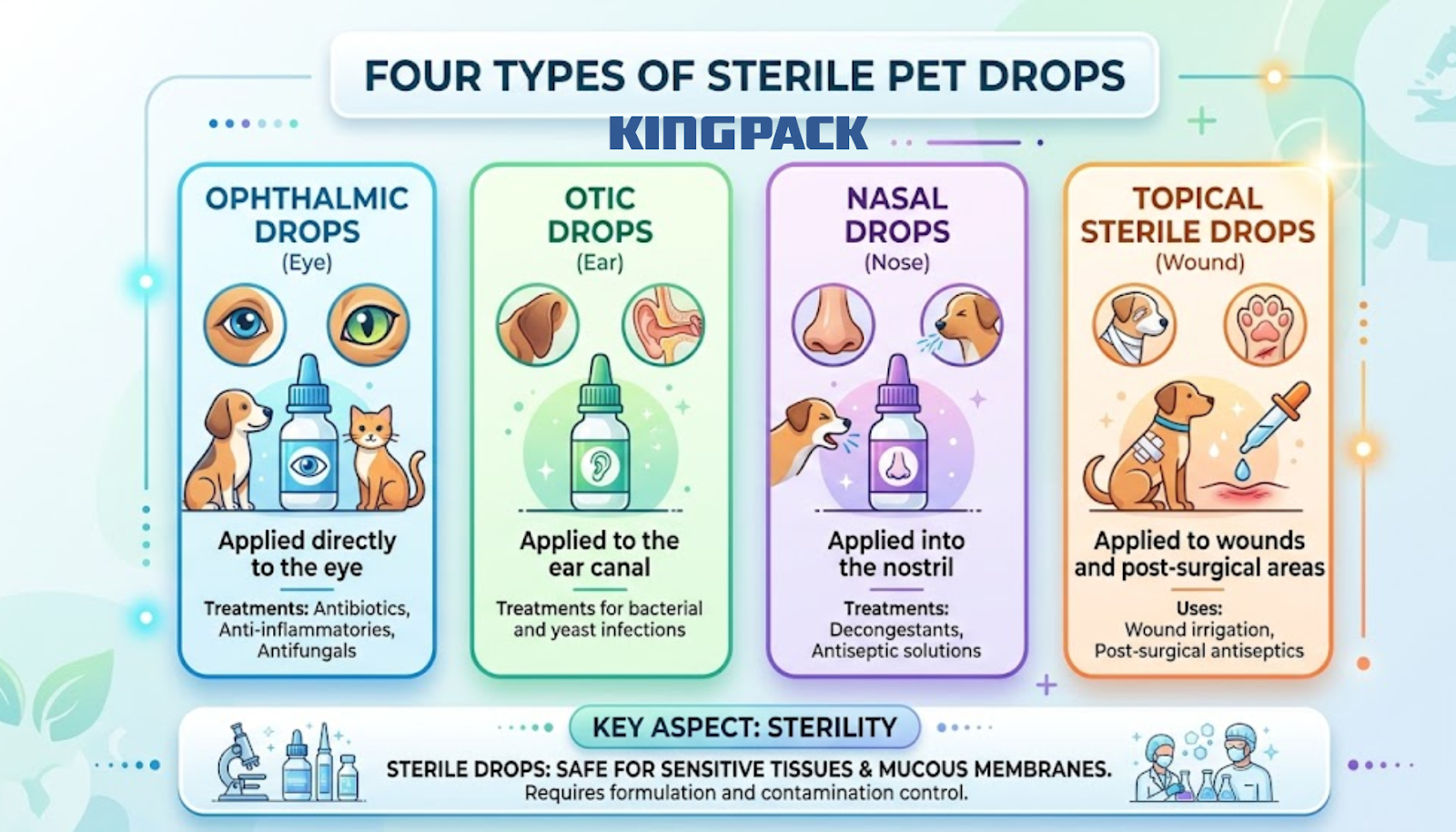
Sterile pet pharmaceutical drops cover four main product categories, each with specific formulation and contamination control requirements:
- Ophthalmic drops: antibiotics, anti-inflammatories, and antifungal treatments applied directly to the eye
- Otic drops: ear canal treatments for bacterial and yeast infections
- Nasal drops: decongestants and antiseptic solutions
- Topical sterile drops: wound irrigation and post-surgical antiseptic applications
Each of these products makes direct contact with sensitive tissue or mucous membranes. According to Accio, in ophthalmic applications specifically, contaminated ophthalmic solutions can lead to serious health repercussions, such as ocular infections, which may result in complications like vision loss.
The same principle applies to veterinary ophthalmic production. A contaminated batch reaching an animal patient is a clinical failure, a regulatory event, and a liability.
Risks of Microbial Contamination in Veterinary Applications
Aseptic processing is critical to maintain product integrity and patient safety when handling sterile liquids in veterinary manufacturing. Contamination can arise from personnel, equipment surfaces, raw materials, the environment, and primary packaging.
The highest risk occurs during the filling operation, where the product is exposed to the environment. Every moment of open exposure increases the chance of contamination. To protect both the batch and the patients, the fill zone must operate under good manufacturing practices and highest quality standards, maintaining Grade A conditions throughout the fill cycle.
Implementing these measures also helps reduce waste and improve efficient handling of batch sizes, ensuring that each batch meets regulations and maintains safety and quality for veterinary patients.
Regulatory Expectations for Pharmaceutical Companies in Veterinary Sterile Production
For the manufacture of sterile products there are four grades of cleanroom zone. According to Precedence Research, Grade A is the critical zone for high-risk operations such as the aseptic processing line, filling zone, stopper bowl, open primary packaging, or for making aseptic connections under the protection of first air.
EU GMP Annex 1, WHO TRS 1044, and FDA cGMP guidance all apply the same grading logic to veterinary sterile manufacturing. Specific guidance regarding terminally sterilized veterinary medicinal products can be found within Annex 4 of the GMP guidelines. Manufacturers supplying export markets need to design their GMP-compliant filling line to satisfy the most stringent applicable framework, not just the minimum domestic requirement.
Recommended Reading: Tube Packaging Solutions in the Pet Care Industry: King Pack’s Advanced Manufacturing Standards – King Pack Machinery
Overall Architecture of a GMP-Compliant Sterile Filling Line
A complete sterile dropper bottle filling line for veterinary drops is not a single machine. It is a connected system covering four processing stages, each of which must be designed and validated as part of the overall aseptic process.
Water Treatment and Sterile Utility Systems Operational Efficiency
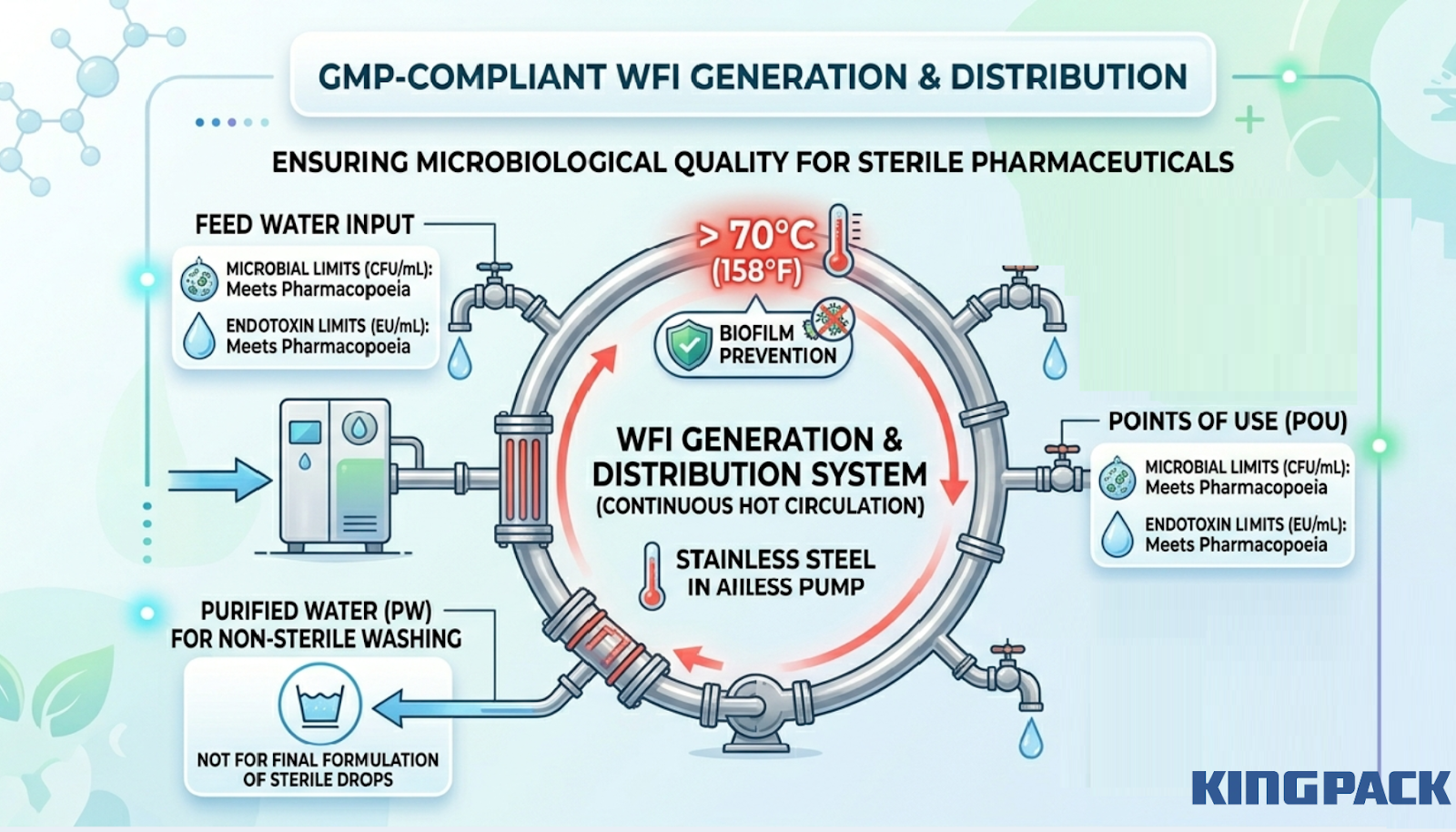
Water for Injection (WFI) is the primary solvent in most aqueous veterinary sterile drops. The WFI generation and distribution system must be designed to prevent biofilm formation, maintain temperature above 70°C during continuous circulation, and deliver water that meets pharmacopoeial endotoxin and microbial limits at every point of use.
Purified Water (PW) is acceptable for some non-sterile washing steps but is not appropriate for the final formulation of sterile ophthalmic or injectable drops. The utility system design determines the microbiological quality of every downstream step.
Sterile Solution Preparation and Filtration
Formulation is typically carried out in a Grade C or Grade D environment, depending on the bioburden risk of the formulation process. The prepared solution is then sterile-filtered through a 0.22 µm sterilizing-grade filter before transfer to the sterile filling vessel. Marketreportsworld studies shows that gases used in aseptic processes should be filtered through a sterilizing-grade filter with a nominal pore size of a maximum of 0.22 µm at the point of use.
Filter integrity testing before and after filtration is a batch release requirement, not an optional quality check.
Aseptic Filling Systems and Stoppering Module
This is the most critical stage of the entire process. The sterile veterinary filling machine dispensing the product into dropper bottles or vials must operate entirely within a Grade A environment. The Grade A area is dedicated to high-risk operations such as fill and finish, stopper bowls, open ampoules, vials, and making aseptic connections.
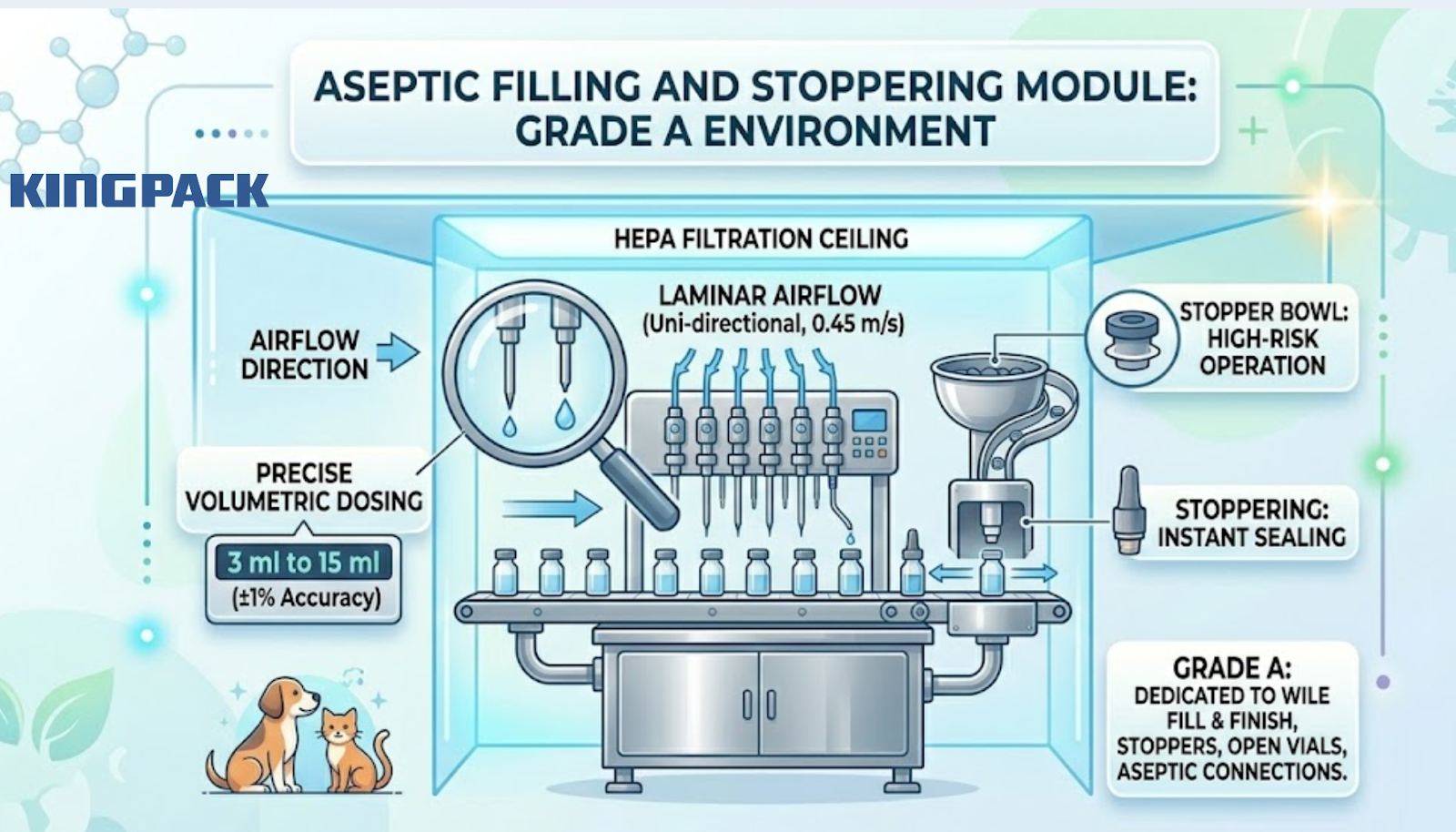
For small-volume pet pharmaceutical drops, fill volumes typically range from 3 ml to 15 ml. At these volumes, a laminar airflow filling system with precise volumetric dosing is required to hit ±1% accuracy consistently at production speed.
Capping, Sealing and In-Line Inspection in Aseptic Filling Machines
Immediately after filling and stoppering, containers move to capping and sealing within the controlled environment. In-line inspection covers fill volume verification, container integrity, and visible particulate detection. Automated inspection systems reduce the reliance on manual visual checks, which are a source of human variability and a contamination risk if operators must intervene in the fill zone.
Cleanroom Design and Contamination Control Strategy
Cleanroom Classification and Zoning Layout
ISO Class 5 is required at the critical point of sterile product exposure, such as during aseptic filling, sterile filtration, or vial stoppering. ISO Class 7 is used as a buffer zone surrounding the ISO 5 environment, maintaining airflow and cleanliness to protect the sterile core.
The cleanroom zoning layout for a sterile pet pharmaceutical drops filling line typically follows this structure:
| Zone | GMP Grade | ISO Equivalent | Function |
| Filling zone | Grade A | ISO 5 | Aseptic filling, stoppering, sealing |
| Background zone | Grade B | ISO 5 at rest / ISO 7 in operation | Supports Grade A aseptic operations |
| Preparation zone | Grade C | ISO 7 | Solution preparation, component staging |
| Support zone | Grade D | ISO 8 | Gowning, material entry, equipment cleaning |
Laminar Airflow and HEPA Filtration
The Grade A fill zone is maintained by unidirectional laminar airflow delivered through HEPA filters. Airflow velocity and direction must be validated to demonstrate that contamination cannot migrate from lower-grade zones into the critical fill area. According to Marketreportsworld, airflow visualization studies should demonstrate airflow patterns within cleanrooms and zones, proving that there is no ingress from lower-grade to higher-grade areas.
Personnel Flow vs Material Flow Separation
One of the most common layout errors in sterile filling facility design is allowing personnel and material flows to intersect. Personnel entering the Grade B area follow a defined gowning route through airlocks. Materials enter through separate pass-through hatches with interlocked doors. The two routes must never share the same airlock or access point, because personnel movement is the highest single source of particle generation in a cleanroom environment.
Environmental Monitoring and Microbial Control
A validated environmental monitoring program runs continuously during production. It covers airborne particle counts, viable air sampling, surface contact plates, and personnel glove prints. Results are reviewed against action and alert limits before each batch is released. Any exceedance triggers an investigation before the next production run.
Recommended Reading: Cross-Contamination Prevention: The Gold Standard for Multi-Formula Pet Drop Production – King Pack – King Pack Machinery
Precision Filling Technology for Micro-Volume Pet Drops
Peristaltic Pump vs Piston Pump Selection
The choice of filling mechanism for a sterile pet pharmaceutical drops line depends on three factors: formulation viscosity, required fill accuracy, and sterility assurance requirements.
| Pump Type | Best Application | Accuracy | Sterility Advantage |
| Peristaltic Pump | Aqueous sterile drops, small batch production | ±1% to ±2% | Product contacts only tubing, not pump mechanism |
| Servo Piston Pump | Viscous veterinary formulations, high-speed lines | ±0.5% to ±1% | Full CIP/SIP integration possible |
| Time-Pressure System | Low-viscosity aqueous solutions at high speed | ±1% to ±3% | Simple design, limited viscosity range |
For aqueous ophthalmic and otic veterinary drops, peristaltic pumps offer a meaningful sterility advantage. The product contacts only the inner surface of a replaceable sterile tube. The pump mechanism itself never touches the formulation. This simplifies cleaning validation and reduces the risk of particulate contamination from metal pump components.
For more viscous veterinary formulations, or for lines requiring ±1% accuracy at speeds above 3,000 units per hour, servo piston filling systems deliver better dosing precision. Our pharmaceutical and medical filling lines at King Pack support both configurations, selected based on the specific formulation and production volume requirements of each project.
Achieving ±1% Filling Accuracy in Small-Dose Applications
Sterile pet pharmaceutical drops are typically filled in volumes between 3 ml and 15 ml. At the lower end of that range, filling accuracy demands are proportionally tighter. A 0.05 ml deviation on a 5 ml fill represents 1% of the total dose. At 3 ml, the same absolute deviation represents 1.7%.
Servo-driven dosing systems address this through digital stroke control. The fill volume is set as a parameter, verified against a gravimetric calibration standard, and locked as a batch recipe. In-process weight checks at defined intervals during the fill run confirm that the delivered volume stays within tolerance throughout the batch.
Bubble Prevention and Anti-Drip Nozzle Design
Bubbles in sterile ophthalmic or otic drops are not just an aesthetic problem. They displace fill volume, cause underfill events that fail label claim, and can introduce air-borne contamination into an otherwise sterile fill.
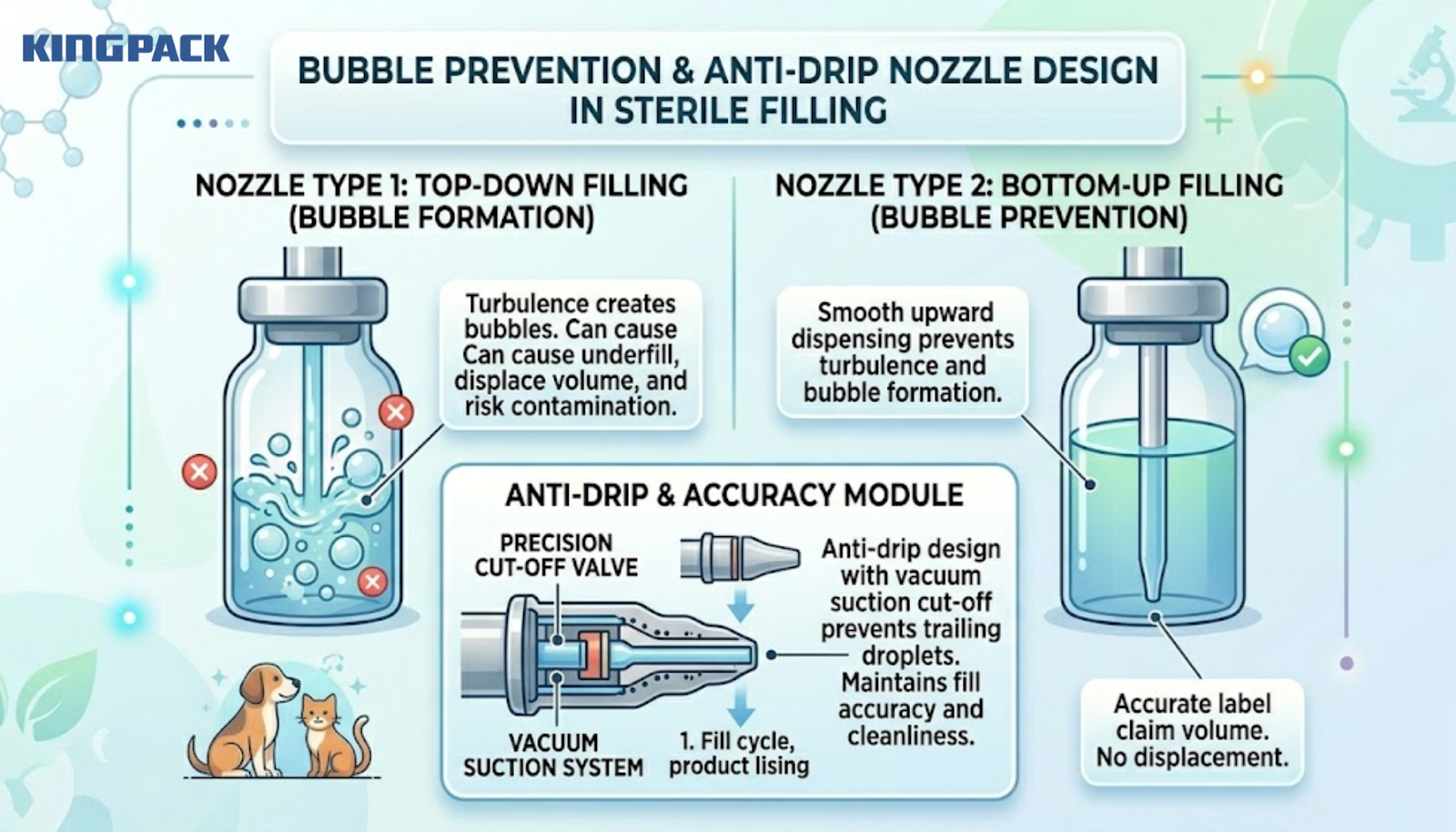
Bottom-up filling, where the nozzle descends into the container and dispenses from the base upward, prevents bubble formation by avoiding the turbulence that top-down filling creates. Anti-drip nozzle design with vacuum suction cut-off prevents trailing droplets between fill cycles, maintaining both fill accuracy and container cleanliness.
Handling Different Viscosities in Veterinary Formulations
Veterinary sterile drops cover a wide viscosity range. Aqueous eye drops for animals may contain viscosity-modifying agents like hydroxypropyl methylcellulose, which significantly increases flow resistance compared to simple saline-based formulations. Oil-based ear drops used in veterinary otic treatment carry viscosities comparable to the anti-parasitic spot-on formulations we discussed in our previous articles.
The filling system must be validated specifically for the viscosity range of each product it handles. Pump selection, nozzle diameter, fill speed, and pipeline pressure all require adjustment when viscosity changes significantly between products on the same line.
Recommended Reading: How to Achieve ±1% Filling Accuracy for High-Value Pet Spot-on Treatments – King Pack Machinery
CIP and SIP Integration for Sterile Operation
Automatic Cleaning-in-Place Systems
Manual cleaning of wetted components on a sterile filling line introduces two risks: incomplete cleaning due to operator variability, and microbial contamination introduced during the cleaning process itself. CIP systems eliminate both by automating the cleaning sequence with validated parameters.
A CIP cycle for a sterile veterinary filling line typically includes a water rinse, a detergent wash at defined concentration and temperature, a second rinse, and a final WFI flush. The sequence, dwell times, flow rates, and temperatures are all validated and recorded automatically. The batch record includes the CIP cycle data as part of the cleaning verification for each production run.
Steam-in-Place Sterilization of Product Contact Parts
SIP follows CIP on a sterile line. Saturated steam at 121°C is circulated through all wetted pathways for a validated dwell time, typically 30 minutes or more depending on the system volume and thermal validation data. Temperature sensors at the coolest points in the system confirm that sterilization conditions were achieved throughout.
SIP eliminates the need to disassemble the filling head for autoclave sterilization between batches, which significantly reduces the manual intervention in the sterile zone and the associated contamination risk.
Reducing Manual Intervention and Cross-Contamination Risks
Every manual action inside the Grade A fill zone is a contamination event waiting to happen. Automated CIP and SIP, servo-controlled filling, automatic container handling, and in-line inspection all reduce the number of times a person must enter or interact with the sterile fill environment.
Where human intervention cannot be eliminated, isolator technology or Restricted Access Barrier Systems (RABS) physically separate the operator from the fill zone. We design our aseptic filling lines for veterinary drugs with RABS integration as a standard option, and full isolator compatibility for manufacturers requiring the highest sterility assurance level.
Recommended Reading: Packaging Machine Solutions – King Pack Packaging Equipment – King Pack Machinery
Validation and Qualification Requirements
IQ, OQ, PQ for Veterinary Sterile Filling Lines
Validation of a GMP-compliant filling line for sterile pet pharmaceutical drops follows a three-stage qualification structure. Installation Qualification confirms that the equipment was installed according to design specifications. Operational Qualification verifies that the equipment performs within defined parameters across its operating range. Performance Qualification demonstrates consistent performance under actual production conditions across multiple runs.
Each stage generates documented evidence that becomes part of the permanent equipment record and supports batch release for the lifetime of the line.
Media Fill Testing and Aseptic Process Simulation
Media fill testing is the primary method for validating that the aseptic filling process can produce sterile product reliably. A microbiological growth medium is filled in place of the actual product, using the same equipment, personnel, procedures, and environmental conditions as a real production batch. The filled units are incubated and examined for microbial growth.
Regulatory guidelines require that media fills be performed at minimum twice per year per shift configuration, with a contamination rate target of zero units in fills of fewer than 5,000 units. For larger fills, the acceptable contamination rate is no more than 0.1%.
Batch Record Traceability and Data Integrity
Every parameter recorded during a sterile fill batch must be attributable, legible, contemporaneous, original, and accurate. This ALCOA standard applies to electronic records as much as paper records. Our filling systems use PLC-based data logging with electronic batch records that capture fill parameters, environmental monitoring results, CIP and SIP cycle data, and in-process check results in a single traceable batch record.
Engineering Considerations for Long-Term Compliance
Minimizing Dead Volume and Product Loss
For high-cost sterile veterinary formulations, dead volume in the filling system carries the same financial impact we described in our previous article on precision pumps.
On a sterile line, dead volume also creates an additional compliance problem: product held in dead legs within the pipeline cannot be guaranteed sterile after a defined hold time, and may require the entire line to be re-sterilized if production is interrupted.
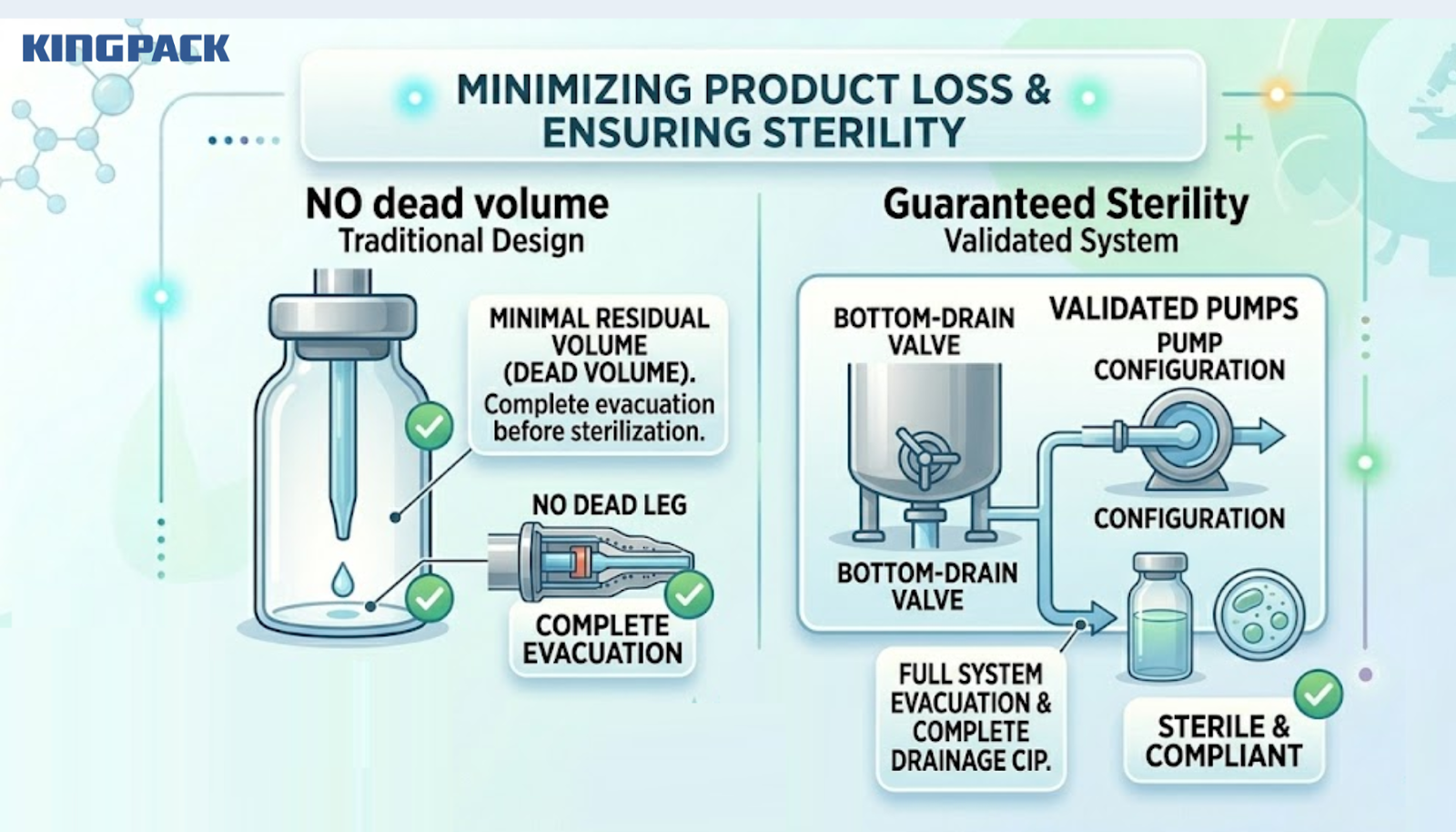
Our sterile filling line designs eliminate dead legs through validated pipeline routing, bottom-drain valve placement, and pump configurations that allow complete system evacuation before CIP initiation.
Modular Design for Future Capacity Expansion
A sterile filling line represents a significant capital investment. Building modularity into the design from the start allows manufacturers to expand capacity without replacing the entire line. Our filling systems use a modular filling head architecture: additional filling stations can be added to an existing line frame without structural changes to the cleanroom or utility connections.
Integration with Labelling, Cartoning and Serialization
The output of the sterile fill line feeds directly into secondary packaging. Label application, cartoning, and serialization systems must be integrated with the fill line data system so that each labelled unit carries a traceable link to its fill batch record. This is a regulatory requirement in most export markets and is increasingly enforced in domestic veterinary pharmaceutical inspections.
Recommended Reading: Tube Packaging Solutions in the Pet Care Industry: King Pack’s Advanced Manufacturing Standards – King Pack Machinery
Common Design Mistakes in Veterinary Sterile Filling Projects
Even experienced manufacturers make avoidable errors when designing a new sterile pet pharmaceutical drops line. The most consequential ones are:
- Underestimating cleanroom airflow requirements, particularly the air change rate needed to maintain Grade B conditions around the Grade A fill zone during active production
- Poor layout planning that forces personnel and material flows to share the same airlock, creating a cross-contamination pathway that is expensive to correct after construction
- Ignoring validation requirements during the design stage, leading to equipment that cannot be qualified without modification
- Selecting an incorrect pump type for the formulation viscosity, producing fill accuracy results during FAT that cannot be reproduced at production speed
- Insufficient automation, leaving too many manual intervention points in the sterile zone that generate both contamination risk and operator fatigue over long production shifts
Why Choose King Pack for Veterinary GMP Filling Lines

Dual-Head Explosion-Proof Liniment Filling and Capping Machine – King Pack Machinery
At King Pack, we engineer GMP-compliant filling lines for sterile pet pharmaceutical drops with full aseptic compliance built into the base design. Our systems are not adapted from general-purpose liquid filling equipment. They are configured from the ground up for the cleanroom environment, sterility assurance requirements, and precision dosing demands of veterinary sterile production.
Our veterinary sterile filling machine configurations include servo-driven piston and peristaltic pump options, RABS and isolator integration, full CIP and SIP circuit design, electronic batch recording, and modular expansion capability. Every system is delivered with a complete validation documentation package covering IQ, OQ, and PQ protocols.
We work with manufacturers at the design stage, before cleanroom construction begins, to review layout, utility requirements, equipment placement, and personnel flow. Getting these decisions right at the design stage costs a fraction of correcting them after installation.
If you are planning a new sterile veterinary filling line or upgrading an existing aseptic filling line for veterinary drugs, contact King Pack for a customized GMP-compliant solution tailored to your specific production requirements.

